Nanodiamonds carry chemotherapy drugs direct to brain tumour
8 October 2013
Microscopic carbon-based particles called nanodiamonds are able to carry chemotherapy drugs directly into brain tumours in a new technique developed by the Jonsson Comprehensive Cancer Center at UCLA.
The new method was found to result in greater cancer-killing efficiency and fewer harmful side effects than existing treatments.
The research has been published in the journal Nanomedicine: Nanotechnology, Biology and Medicine
Glioblastoma is the most common and lethal type of brain tumour. Despite treatment with surgery, radiation and chemotherapy, the median survival time for glioblastoma patients is less than one-and-a-half years. The tumours are notoriously difficult to treat, in part because chemotherapy drugs injected alone often are unable to penetrate the system of protective blood vessels that surround the brain, known as the blood–brain barrier. And those drugs that do cross the barrier do not stay concentrated in the tumour tissue long enough to be effective.
The drug doxorubicin, a common chemotherapy agent, has shown promise in a broad range of cancers, and it has served as model drug for the treatment brain tumours when injected directly into the tumour. Dean Ho of the UCLA School of Dentistry and colleagues from the Lurie Children's Hospital of Chicago and Northwestern University's Feinberg School of Medicine originally developed a strategy for strongly attaching doxorubicin molecules to nanodiamond surfaces, creating a combined substance called ND–DOX.
Nanodiamonds are carbon-based particles roughly 4 to 5 nanometers in diameter that can carry a broad range of drug compounds. The particles have a large accessible surface and tailorable surface chemistry. They also have unique optical, mechanical, and thermal properties and are non-toxic.
Tumour-cell proteins are able to eject most anticancer drugs that are injected into the cell before those drugs have time to work, but they can't get rid of the nanodiamonds. Thus, drug–nanodiamond combinations remain in the cells much longer without affecting the tissue surrounding the tumour.
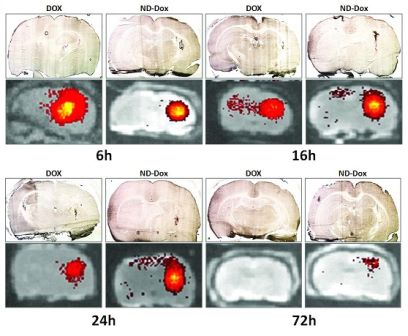
ND-DOX in the brain. These images show the retention of
doxorubicin and ND-DOX in brain tissue, with light microscopic
images (upper rows) and fluorescence images detecting
fluorescence generated from doxorubicin (lower rows). The images
show the distribution of unmodified doxorubicin and ND-DOX after
convection-enhanced delivery (CED) at 6, 16, 24 and 72 hours.
Credit: Jonsson Comprehensive Cancer Center, UCLA
HHo and his colleagues hypothesized that glioblastoma might be efficiently treated with a nanodiamond-modified drug by using a direct-injection technique known as convection-enhanced delivery, or CED. They used this method to inject ND–DOX directly into brain tumors in rodent models.
The researchers found that ND–DOX levels in the tumours were retained for a duration far beyond that of doxorubicin alone, showing that doxorubicin was taken into the tumour and remained their longer when attached to anodiamonds. In addition, ND–DOX was also found to increase apoptosis — programmed cancer-cell death — and to decrease cell viability in glioma (brain cancer) cell lines.
The results also demonstrated for the first time that the ND–DOX delivery limited the amount of doxorubicin that was distributed outside the tumour. This reduced toxic side effects and kept more of the drug in the tumour for longer, increasing the drug's tumour-killing efficiency without affecting the surrounding tissue. Survival time increased significantly in the rats treated with ND–DOX, compared with those given only unmodified doxorubicin.
NNanodiamonds have many facets, almost like the surface of a soccer ball, and can bind to doxorubicin very strongly and quickly, Ho noted. Further research will expand the list of brain-cancer chemotherapy drugs that can be attached to the nanodiamond surfaces to improve treatment and reduce side effects.
"Nanomaterials are promising vehicles for treating different types of cancer," Ho said. "We're looking for the drugs and situations where nanotechnology actually helps chemotherapy function better, making it easier on the patient and harder on the cancer."
This study showed that convection-enhanced delivery of ND–DOX offers a powerful treatment delivery system against these very difficult and deadly brain tumours, Ho said.