Image guided surgical system treats blood clots in the brain
19 August 2013
A new image-guided surgical system under development at Vanderbilt University uses steerable needles to penetrate the brain with minimal damage and suction away blood clots.
For the last four years, the team at Vanderbilt has been developing a steerable needle system for transnasal surgery: operations to remove tumours in the pituitary gland and at the skull base that traditionally involve cutting large openings in a patient’s skull and/or face. Studies have shown that using an endoscope to go through the nasal cavity is less traumatic, but the procedure is so difficult that only a handful of surgeons have mastered it.
The system is described in an article accepted for publication in the journal IEEE Transactions on Biomedical Engineering. It is the product of an ongoing collaboration between a team of engineers and physicians headed by Assistant Professor Robert J. Webster and Assistant Professor of Neurological Surgery Kyle Weaver.
Professor Webster’s design, which he calls an active cannula, consists of a series of thin, nested tubes. Each tube has a different intrinsic curvature. By precisely rotating, extending and retracting these tubes, an operator can steer the tip in different directions, allowing it to follow a curving path through the body.
The system for treating brain-clots only needs two tubes: a straight outer tube and a curved inner tube. Both are less than one twentieth of an inch in diameter. When a CT scan has determined the location of the blood clot, the surgeon determines the best point on the skull and the proper insertion angle for the probe. The angle is dialled into a fixture, called a trajectory stem, which is attached to the skull immediately above a small hole that has been drilled to enable the needle to pass into the patient’s brain.
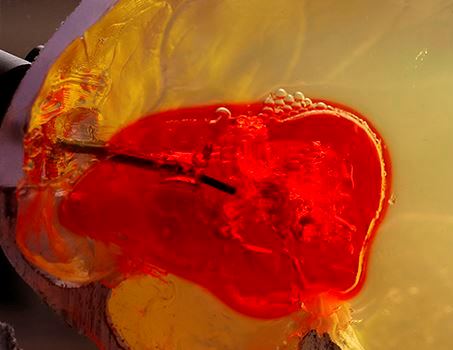
Test of steerable needle system on a phantom
brain clot made from gelatin.
The surgeon positions the robot so it can insert the straight outer tube through the trajectory stem and into the brain. He also selects the small inner tube with the curvature that best matches the size and shape of the clot, attaches a suction pump to its external end and places it in the outer tube.
Guided by the CT scan, the robot inserts the outer tube into the brain until it reaches the outer surface of the clot. Then it extends the curved, inner tube into the clot’s interior. The pump is turned on and the tube begins acting like a tiny vacuum cleaner, sucking out the material. The robot moves the tip around the interior of the clot, controlling its motion by rotating, extending and retracting the tubes. According to the feasibility studies the researchers have performed, the robot can remove up to 92% of simulated blood clots.
“The trickiest part of the operation comes after you have removed a substantial amount of the clot. External pressure can cause the edges of the clot to partially collapse making it difficult to keep track of the clot’s boundaries,” said Webster.
The goal of a future project is to add ultrasound imaging combined with a computer model of how brain tissue deforms to ensure that all of the desired clot material can be removed safely and effectively.