Breakthrough in understanding cell processes could lead to new medical treatments
22 November 2012
Researchers at Tokyo Institute of Technology and the Weizmann Institute of Science have identified a means of controlling biological processes that could help treatments for immune disease, neurological disorders and cancer.
The cellular response to a number of signals including inflammatory cytokines, tumour promoters, carcinogens, and chemotherapeutic agents hinge on the transcription factor NF-κB.
Rivka Dikstein and colleagues at Tokyo Institute of Technology in Japan and the Weizmann Institute of Science in Israel have identified how the protein DSIF controls NF-κB activity. The research may lead to methods for controlling inflammation, and immune responses, as well as the cell cycle processes that can lead to cancer.
Activity of NF-κB is usually transient because some of the gene products function as negative regulators of NF-κB, resulting in a negative feedback loop. This is desirable because persistent activation of NF-κB can lead to various pathogenic conditions such as chronic inflammation, autoimmune diseases, and cancer.
Dikstein and colleagues treated cells to reduce the expression of DSIF and monitored the effect on protein levels of NF-κB’s target genes. They found that in the treated ‘DSIF knockdown’ cells the levels of these proteins failed to recover following a degradation process. Further investigation revealed that DSIF plays an integral role in the maturation and transport of messenger RNAs (Figure 2).
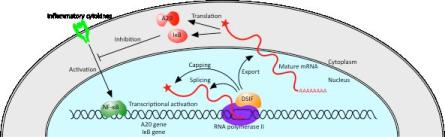
Figure 2. The role of DSIF in the control of
inflammatory responses. Upon NF-κB activation, DSIF is recruited to
the A20 and IκBα genes and contributes to the negative feedback
regulation in a unique manner. DSIF facilitates the expression of
these genes by stimulating capping, splicing, and export of their
mRNAs. Otherwise, these steps are rate-limiting, and A20 and IκBα
proteins are not synthesized successfully.
“The unique control of the negative feedback regulator genes by DSIF may be utilized by cells under circumstances in which prolonged NF-κB activity is needed,” explain the authors. Their future studies will focus on DSIF under specific settings with a view to identifying drug targets for selective manipulation of NF-κB activity.
Background
NF-κB induces gene expression responsible for a number of biological processes including inflammation and cell survival. Its deregulation is linked to chronic inflammation and cancer but so far the mechanisms behind these processes have not been fully understood.
Nuclear transport is integral to NF-κB activity. In unstimulated cells it is retained inactive in the cytoplasm. Signals that trigger its activation result in its transport into the nucleus where it activates responsive genes, such as A20 and IκBα.
Activity of NF-κB is usually transient because products of these genes function as negative regulators of NF-κB, resulting in a negative feedback and limiting the duration of NF-κB activation (Figure 1). In certain diseases NF-κB activation becomes persistent, perhaps due to an interruption of this feedback loop.
NF-κB activation results in recruitment of the protein DSIF at the target genes A20 and IκBα. The researchers confirmed the role of DSIF in the negative feedback regulation of NF-κB activity by downregulating a protein subunit of DSIF and comparing the treated cells with controls. In untreated cells the levels of proteins for the target genes A20 and IκBα are high. Although the levels diminish significantly in 30 minutes following a process called TNF-α induction, which is caused by degradation of the proteins, within 2 hours the levels recover. In knockdown cells, however, the levels remained diminished 2 hours after TNF-α induction.
Further investigations using chromatin immunoprecipitation assays revealed abnormalities in the synthesis of A20 and IκBα proteins in the knock down cells. A significant portion of the A20 and IκBα mRNAs in the knockdown cells were uncapped and unspliced (Fig. 2).
The researchers also studied how export was affected in DSIF knockdown cells. Export of mature mRNAs from the nucleus to the cytoplasm is a highly regulated process incorporating quality assurance checks. The researchers prepared RNA from cytosolic and nuclear cell fractions for comparison. In knockdown cells following TNF-α induction, the nuclear fractions had much larger amounts of A20 and IκBα than the cytosolic fractions, suggesting accumulation in the nucleus.
The impact observed on the synthesis and export of A20 and IκBα in DSIF knockdown cells was striking. A20 and IκBα are also responsible for regulating other cell signalling processes and it is likely that the effects on associated signalling processes contribute to the overall impact of DSIF knockdown.
As the authors point out, diminished DSIF activity could contribute to pathological states in which NF-κB becomes constitutively active, such as inflammatory and autoimmune diseases, neurological disorders, and cancer. Further work on DSIF activity could help develop new approaches to treating these diseases.